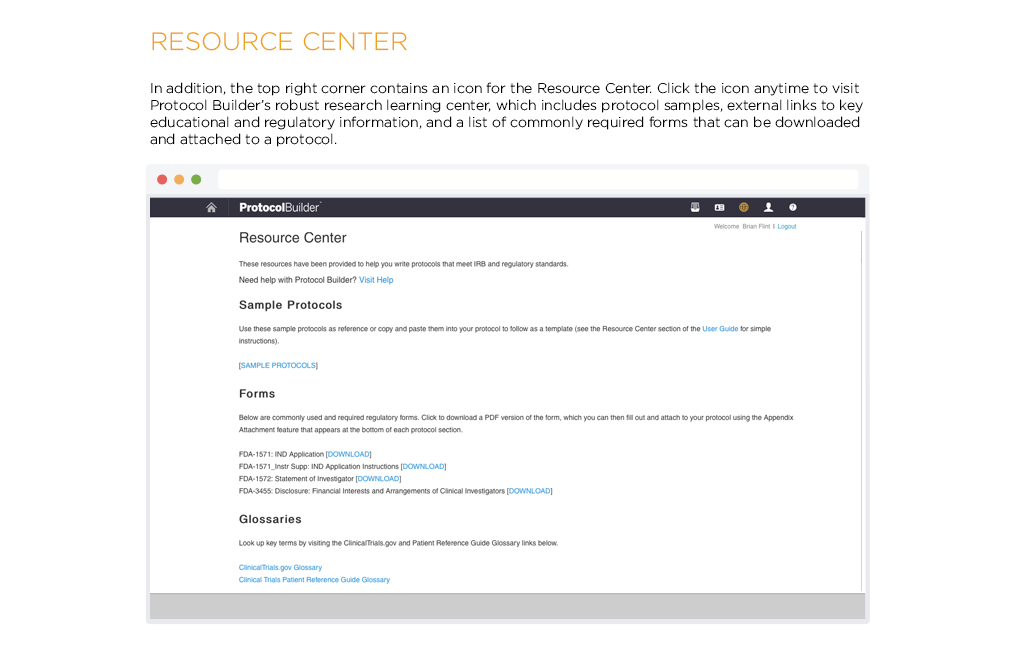
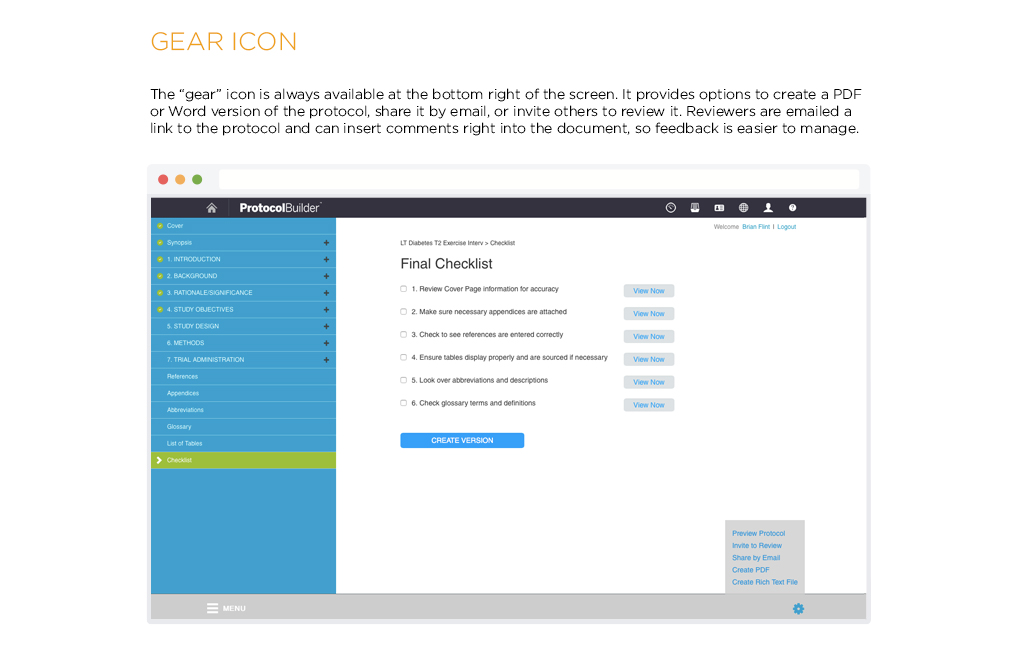
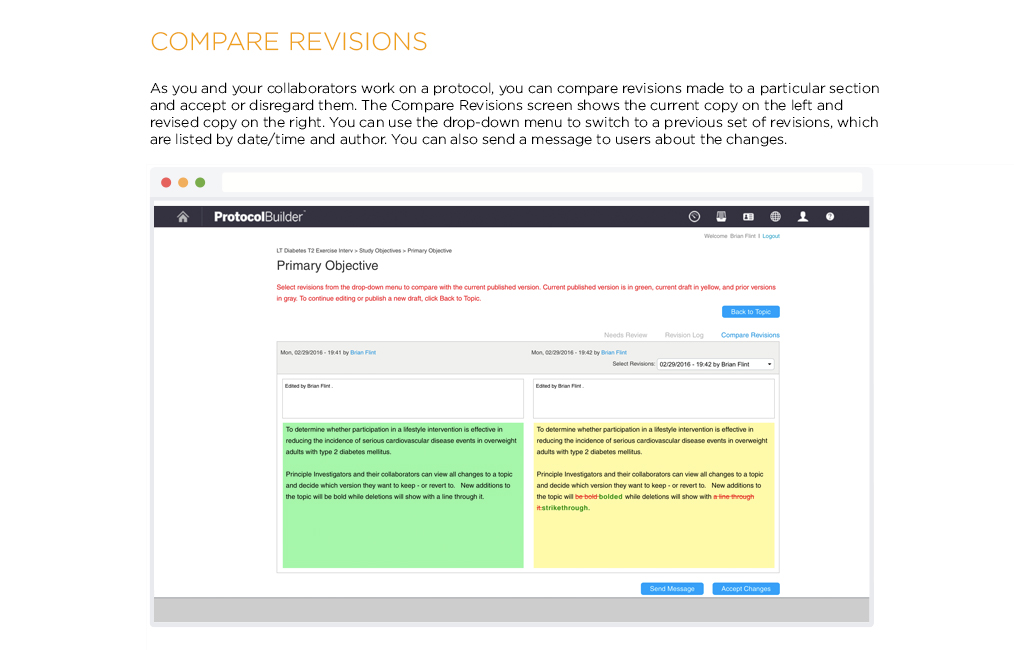
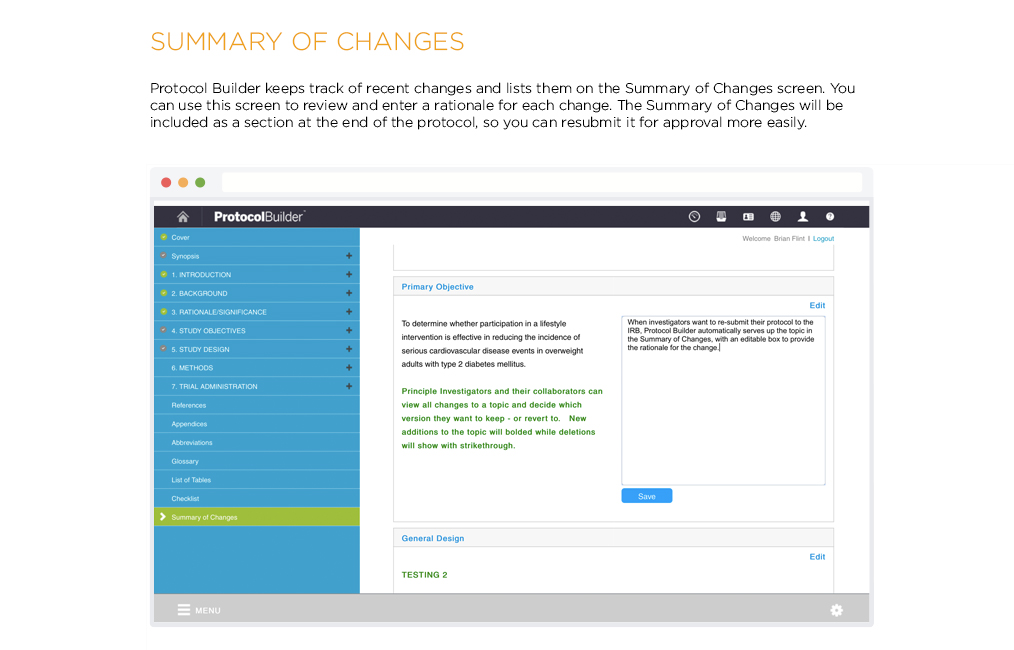
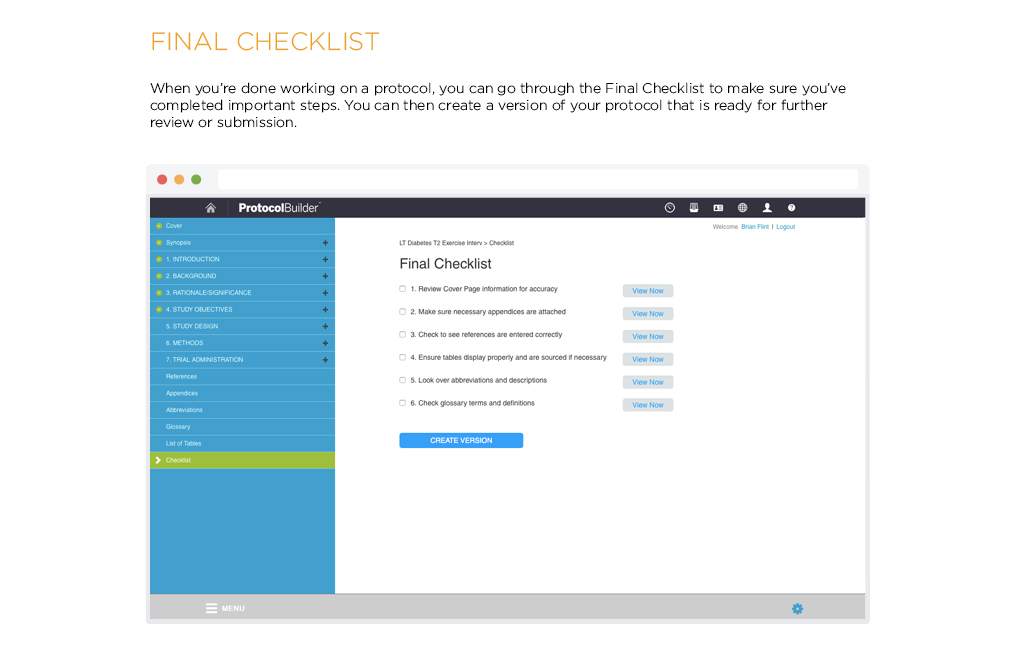
[vc_row css_animation=”” row_type=”row” use_row_as_full_screen_section=”no” type=”full_width” angled_section=”no” text_align=”left” background_image_as_pattern=”without_pattern” css=”.vc_custom_1504633514106{margin-right: 60px !important;margin-left: 60px !important;}”][vc_column css=”.vc_custom_1410463246763{margin-top: 0px !important;margin-bottom: 60px !important;}”][vc_column_text]
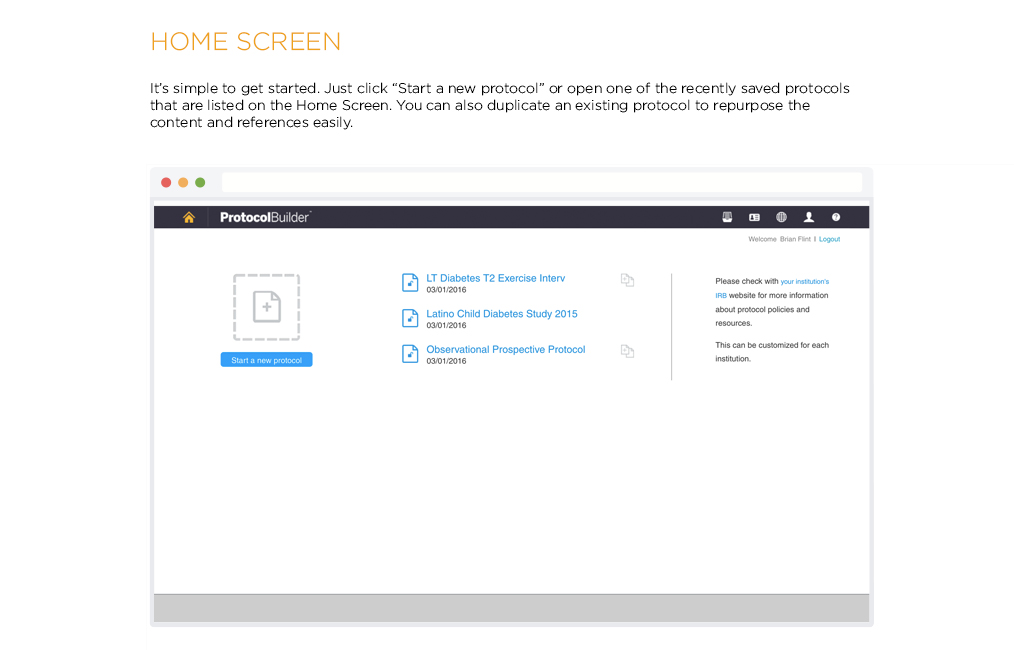
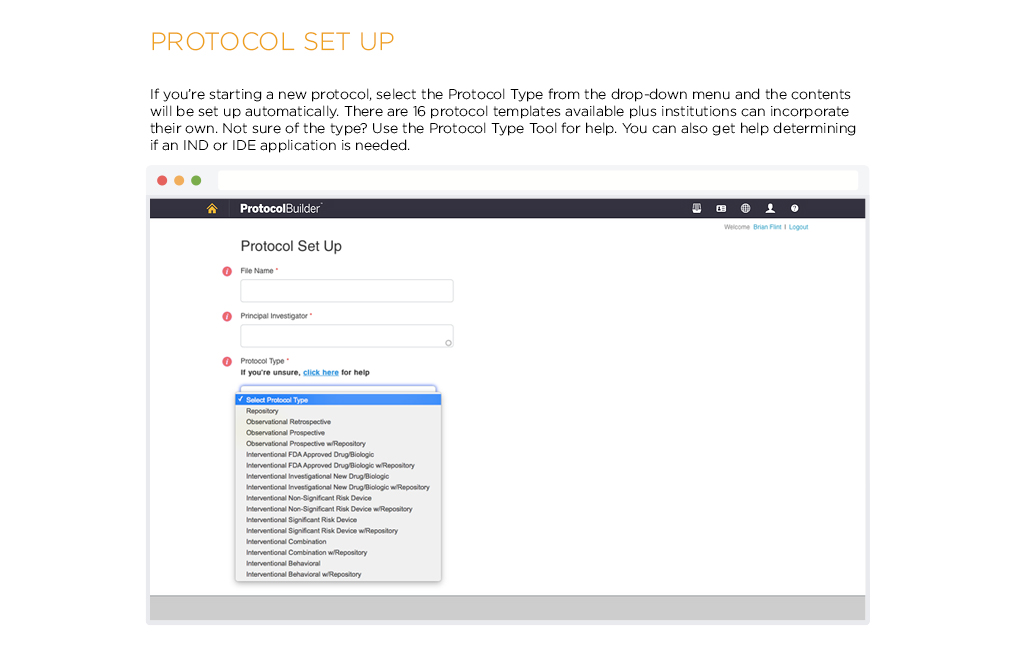
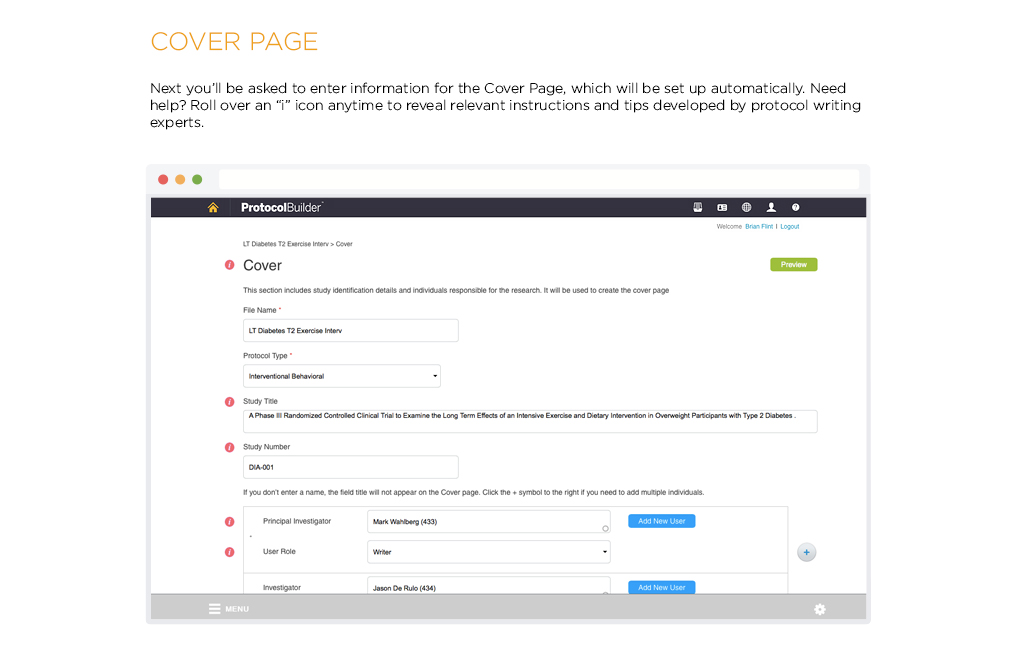
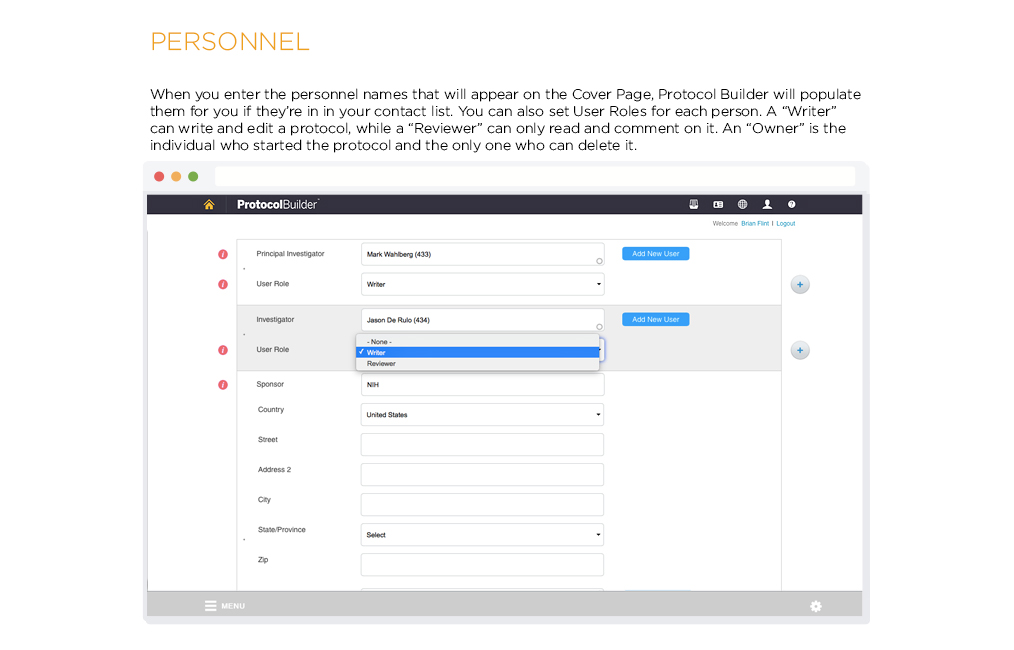
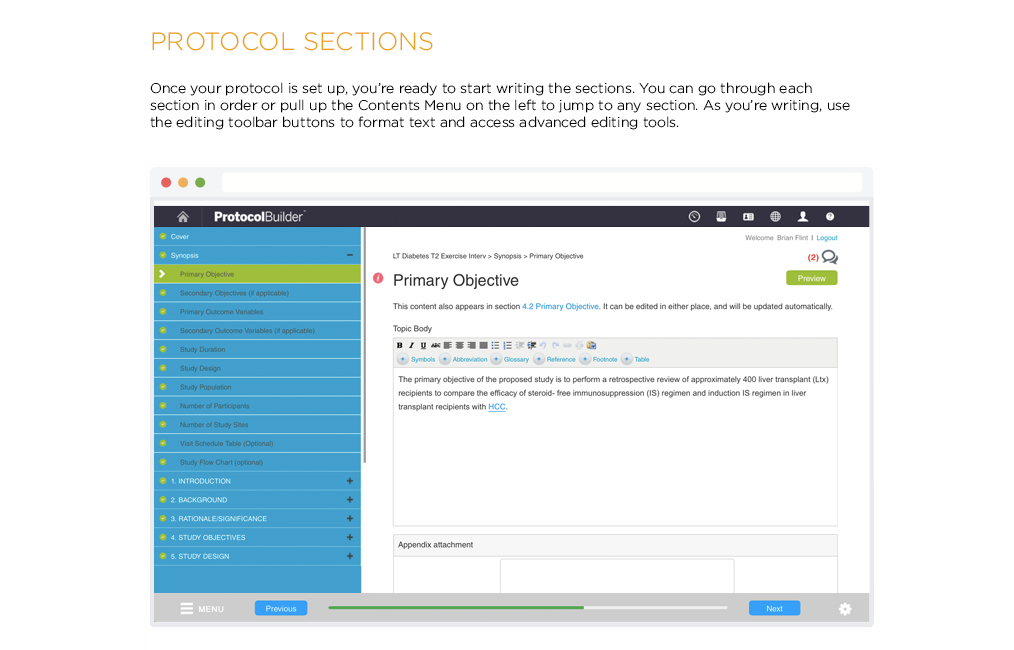
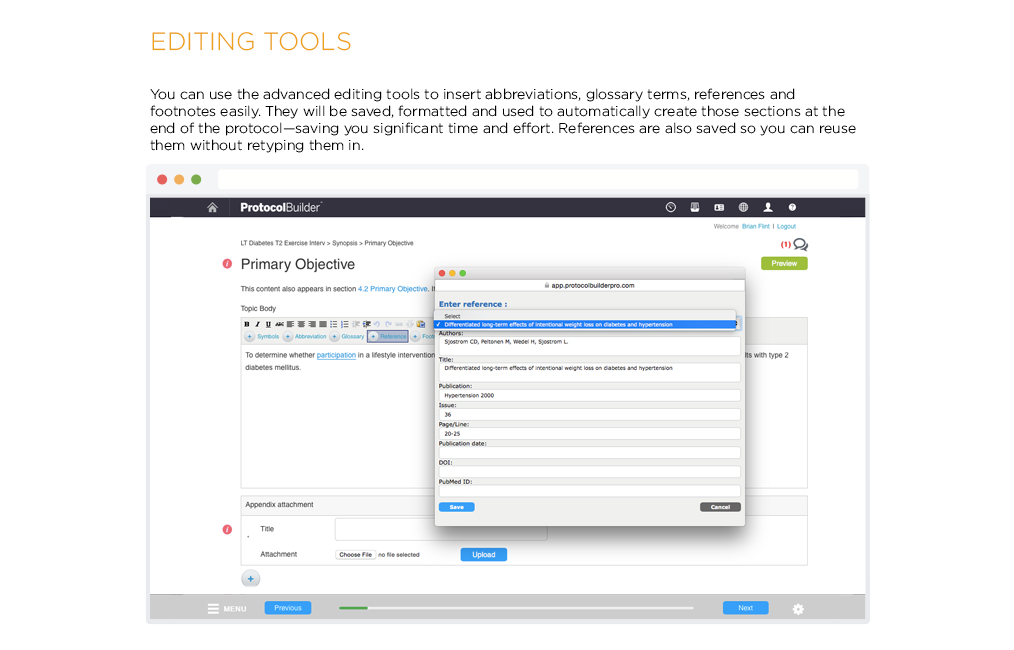
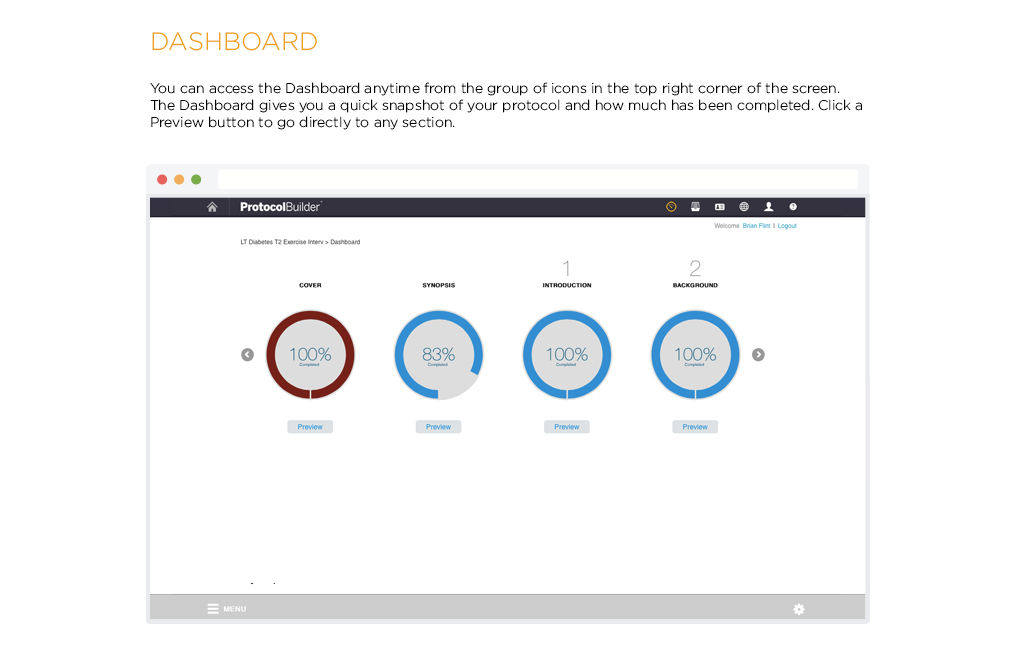
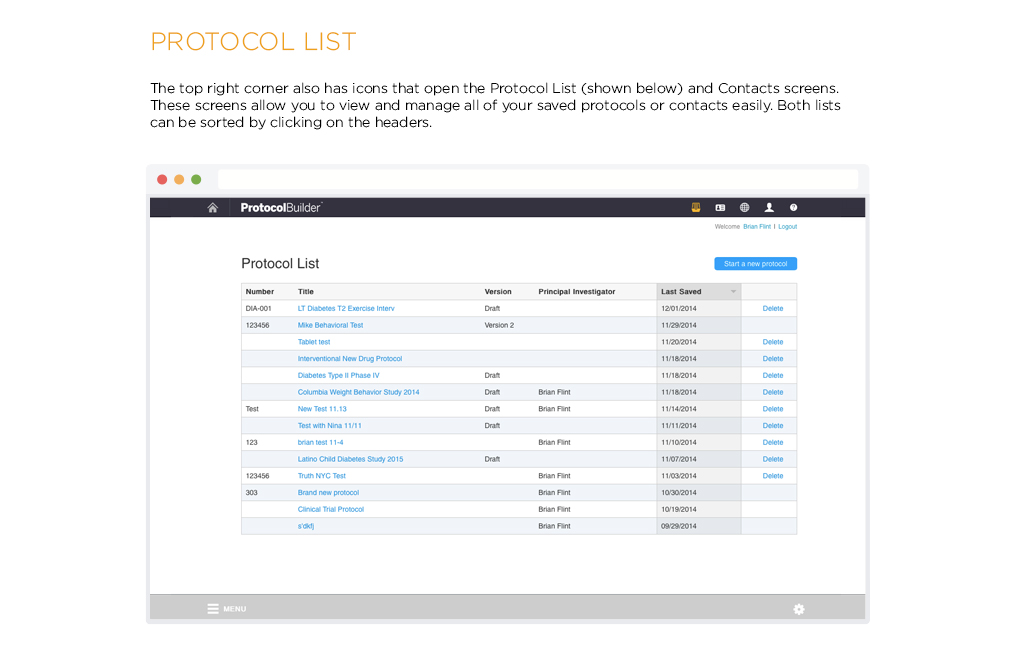
Time Saving Features
[/vc_column_text][vc_column_text el_class=”make-margin-bottom-fifteen”]
Carefully designed features help improve the quality and consistency of clinical research protocols to make internal and IRB review processes more efficient.
[/vc_column_text][vc_empty_space][vc_row_inner row_type=”row” type=”full_width” text_align=”left” css_animation=””][vc_column_inner el_class=”make-margin-top-sixty-six” width=”1/4″][homepage_icons_stylethree textalign=”right” icontype=”icon-code” title=”Automatic Set Up” text=”Enter basic information and appropriate sections are set up automatically or duplicate an existing protocol to repurpose. Get help determining the type of study and whether or not you need an IND or IDE application.”][homepage_icons_stylethree textalign=”right” icontype=”icon-tools” title=”Advanced Editing Tools” text=”Insert references, tables, appendices, abbreviations and glossary terms easily as you write. Those sections are then set up automatically at the end of the document.”][homepage_icons_stylethree textalign=”right” icontype=”icon-menu” title=”Contents Menu” text=”Go step-by-step through the writing process or use the menu to navigate to any section. Switch to the preview mode to read through it more easily.”][/vc_column_inner][vc_column_inner width=”1/2″][vc_single_image image=”2418″ img_size=”585×707″ alignment=”center” qode_css_animation=””][/vc_column_inner][vc_column_inner el_class=”make-margin-top-sixty-six” width=”1/4″][homepage_icons_stylethree icontype=”icon-compass” title=”Expert Guidance” text=”See simple instructions and get relevant tips developed by experts as you go. You’ll also find sample investigator-initiated protocols in the Resource Center that you can use as a template.”][homepage_icons_stylethree icontype=”icon-archive” title=”Resource Center” text=”Access well-written samples as well as links to important educational and regulatory information. A list of commonly used forms is also available for download. “][homepage_icons_stylethree icontype=”icon-share” title=”Collaboration and Change Tracking Tools” text=”Invite others to review, edit, or add comments to a protocol. Compare prior revisions to current content and see a history of all changes. Go back to previously versions anytime.”][/vc_column_inner][vc_column_inner][vc_empty_space][/vc_column_inner][vc_column_inner el_class=”!important; text-align:center;” css=”.vc_custom_1409685711594{padding-right: 200px !important;padding-left: 200px !important;}”][vc_column_text el_class=”BLUE BUTTON”]
Download the Time Saving Sheet (PDF) to see how Protocol Builders eliminate or streamline time-consuming tasks.
[/vc_column_text][vc_empty_space][vc_column_text el_class=”BLUE BUTTON”]
Start your free trial to see the benefits for yourself.
[/vc_column_text][/vc_column_inner][vc_column_inner][vc_empty_space height=”80px”][/vc_column_inner][/vc_row_inner][vc_empty_space][/vc_column][/vc_row][vc_row css_animation=”” row_type=”row” use_row_as_full_screen_section=”no” type=”full_width” angled_section=”no” text_align=”left” background_image_as_pattern=”without_pattern”][vc_column][vc_column_text]
[/vc_column_text][/vc_column][/vc_row]